Introduction
Measurable residual disease (MRD) monitoring is predictive in acute myeloid leukemia (AML). Assessment of FMS3-like tyrosine kinase-3 in-frame internal tandem duplications (FLT3-ITD) is usually performed at diagnosis by polymerase chain reaction (PCR). Due to its relative lower sensitivity of 2%, FLT3-ITD PCR is not routinely used during response assessment. Next-generation sequencing is similarly limited by technical difficulties in capturing tandem duplications using the short base pair-based sequencing method. Long base pair-based sequencing has been reported, but its use is limited by financial restrictions. Multiparameter flow cytometry (MFC) can be a useful tool for MRD monitoring in this AML subtype until such time that molecular techniques for detecting FLT3-ITD MRD are optimized.
Patients and methods
The study evaluated the outcomes of FLT3-ITD mutated AML patients diagnosed and treated from 2018 to 2022 at Princess Margaret Cancer Centre. We compared outcomes according to MFC-MRD post-induction and FLT3-ITD allele frequency (AF) status at diagnosis. MRD cut-off was 0.1%. Data were locked as of June 30, 2023. Clinical outcomes evaluated include overall survival (OS) and relapse-free survival (RFS). The cumulative incidence of relapse (CIR) and non-relapse mortality (NRM) were calculated considering competing risk. The Kaplan-Meier method using a log-rank test and a multivariate Cox proportional hazard model was used for analyses, while the Gray test and Fine-Grey model were used for uni- and multivariate analysis for CIR and NRM.
Results
A total of 111 patients with a mean age of 63.5 years were included, of whom 90 received treatment. Secondary AML accounted for 12.7% of patients. Risk stratification according to European LeukemiaNet (ELN) 2022 was favorable in 2 (1.8%), intermediate in 67 (60.4%), and adverse in 42 patients (37.8%). Nucleophosmin 1 (NPM1) co-mutation was observed in 55 patients (49.5%). Seventy-nine patients (87.8%) could be assessed for overall response, including 69 (76.7%) who achieved complete remission (CR) or CR with incomplete count recovery (CRi). Of these, 54 achieved first CR/CRi (CR1) with 1 induction cycle. MFC-MRD data were available in 61 patients, of whom 44 (72.1%) were MRD negative, while 17 (27.9%) were MRD positive. With a median follow-up of 437 days, 50 patients (45%) were still alive. Median OS and RFS were 3.42 years and 1.05 years, respectively.
Among patients who achieved CR1, post-induction MFC-MRD positivity correlated with an inferior OS (HR 2.35 [1.06-5.26], p=0.037) and a trend for a shorter RFS (HR 2.08 [0.99-4.35], p=0.052). We examined the impact of FLT3-ITD AF at diagnosis on long-term outcomes. By applying a binary recursive partitioning method, the cut-off of FLT3-ITD AF with the best risk stratification power for RFS, was defined at 54.6%. The group with a higher FLT3-ITD AF showed inferior OS (HR 1.86 [1.01-3.44], p=0.047) and RFS (HR 1.91 [1.09-3.33], p=0.023).
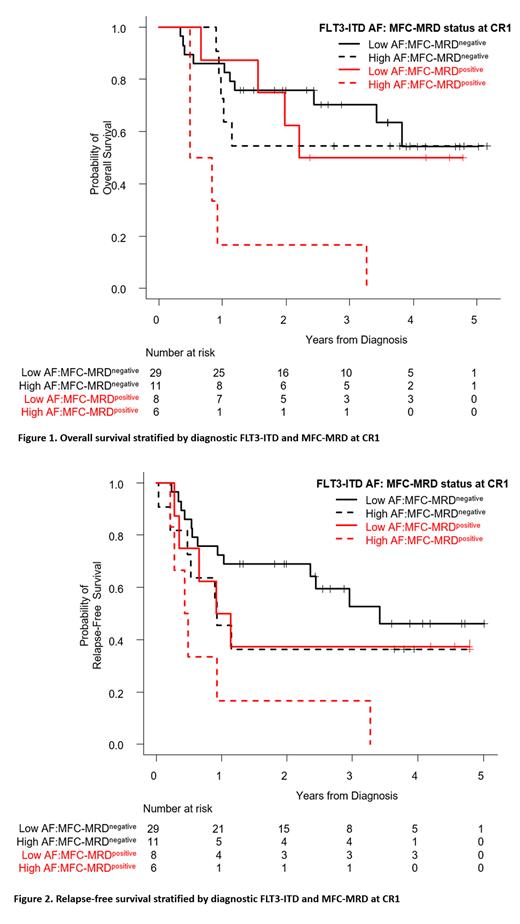
Taking together FLT3-ITD AF at diagnosis and MFC-MRD status at CR1, patients with low FLT3-ITD AF and negative MRD had the highest OS rate of 86.2% at 12 months (p=0.023), and the highest RFS at 72.4% (p=0.096), while the corresponding values for low FLT3-ITD AF/positive MRD patients were 87.5% and 50%, respectively. In contrast, those with high FLT3-ITD AF/negative MRD had an OS of 72.7% and a RFS of 45.5%, while those with high FLT3-ITD AF/positive MRD showed the lowest OS (16.7%) and the shortest RFS (16.7%). There was no statistical difference in CIR and NRM among groups.
Multivariate analysis with stepwise selection was performed, considering age at diagnosis, ELN 2022 risk, MFC-MRD at CR1, FLT3-ITD AF at diagnosis, cytogenetics, and NPM1 co-mutation. Predictive factors for OS were MFC-MRD post-induction (HR 2.49 [1.11-5.61], p=0.027) and FLT3-ITD AF (HR 2.29 [1.03-5.11], p=0.043). For RFS, age at diagnosis (HR 1.03 [1.00-1.06], p=0.038) and FLT3-ITD AF (HR 2.40 [1.15-5.01], p=0.019) were predictive, while MFC-MRD was not significant (HR 1.59 [0.72-3.53], p=0.25).
Conclusion
Our data demonstrate that MFC-based MRD assessment is feasible in AML with FLT3-ITD. Although better outcomes are expected in patients with a lower FLT3-ITD AF, patients who failed to achieve MRD negativity at CR1 showed inferior outcomes. The presence of both poor risk factors, a high AF of FLT3-ITD at diagnosis and MRD positivity at CR1, correlated with the worst treatment outcomes.
Disclosures
Richard-Carpentier:Pfizer: Honoraria, Membership on an entity's Board of Directors or advisory committees; Bristol-Myers Squibb: Honoraria, Membership on an entity's Board of Directors or advisory committees; Taiho: Honoraria, Membership on an entity's Board of Directors or advisory committees; Astellas: Honoraria, Membership on an entity's Board of Directors or advisory committees; AbbVie: Membership on an entity's Board of Directors or advisory committees. Schimmer:UHN: Patents & Royalties: the use of DNT cells to treat AML; Medivir AB: Research Funding; Jazz: Consultancy, Honoraria; Otsuka Pharmaceuticals: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Medical and Scientific Advisory Board of the Leukemia and Lymphoma Society of Canada: Membership on an entity's Board of Directors or advisory committees; BMS: Research Funding; Takeda Pharmaceuticals: Consultancy, Honoraria, Research Funding. Schuh:Amgen: Honoraria, Research Funding; Glycomimetics: Research Funding; Pfizer: Consultancy, Honoraria; Servier: Honoraria, Research Funding; Agios: Honoraria, Research Funding; Astellas: Honoraria, Research Funding; Kite/Gilead: Research Funding; Abbvie: Honoraria, Research Funding; Bristol Myers Squibb: Honoraria, Research Funding; Teva: Consultancy, Honoraria. Chan:Servier: Research Funding; AbbVie: Research Funding; BMS: Research Funding; Agios: Research Funding. Mattsson:Magenta Therapeutics Inc: Consultancy, Honoraria; Takeda Canada Inc: Consultancy, Ended employment in the past 24 months, Honoraria; Merck Canada Inc: Ended employment in the past 24 months, Honoraria, Speakers Bureau; Jazz Pharmaceuticals: Consultancy, Honoraria; Medexus: Honoraria, Other: advisory board; Sanofi Canada: Honoraria, Other: advisory board. Gupta:BMS Celgene, Roche, AbbVie, Pfizer, Sierra Oncology, CTI Biopharma, GSK: Other: Participation on a Data Safety Monitoring Board or Advisory Board; GSK: Other: Travel to EHA 2023 for invited talk at GSK sponsored MPN education session ; Novartis, BMS Celgene, SMP Oncology, AbbVie, Constellation Biopharma, Pfizer, GSK Pharma, CTI Biopharma: Consultancy; Novartis, BMS Celgene, GSK: Honoraria; BMS, Celgene, Roche, Abb Vie, Pfizer, Sierra Oncology, CTI Biopharma: Membership on an entity's Board of Directors or advisory committees; Novartis, BMS Celgene, Sierra Oncology, AbbVie, Constellation Biopharma, Pfizer, GSK Pharma, CTI Biopharma: Consultancy. Tierens:BD Biosciences: Honoraria, Speakers Bureau. Kim:Paladin: Consultancy, Research Funding; Pfizer: Consultancy, Honoraria, Research Funding; BMS: Research Funding; Novartis: Consultancy, Honoraria, Research Funding.